iSTAR Medical’s glaucoma device MINIject shows positive one-year results in European trial
WAVRE, Belgium: iSTAR Medical, a medtech company developing minimally invasive ophthalmic implants for treatment of patients with glaucoma, today announced positive one-year results from the European STAR-II clinical trial of its micro-invasive glaucoma surgery (MIGS) device, MINIjectTM. The results have been presented at the virtual American Academy of Ophthalmology (AAO) annual meeting, held 13-15 November 2020.
Positive one-year results from the STAR-II European trial sustain the remarkable six-month outcomes and are consistent with the STAR-I trial results at one year
Promising MINIject data at two years from the STAR-I trial were recently published in the British Journal of Ophthalmology
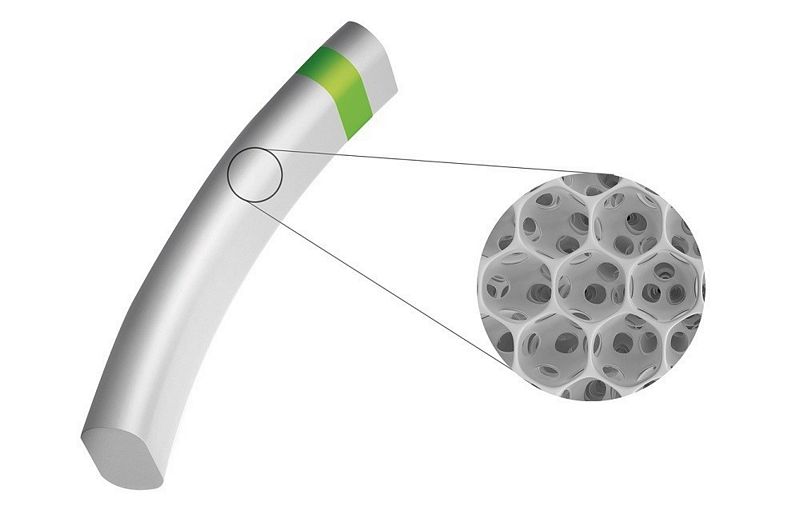
Pre-clinical studies of MINIject highlighting the anti-fibrotic properties of its STAR® material were recently published in BMC Biomedical EngineeringSTAR-II results at one year
At one-year follow-up, mean intraocular pressure (IOP) was 15.1 mm Hg, corresponding to a 38 percent reduction from medicated baseline IOP. Moreover, 45 percent of patients did not require any IOP-lowering medication one year after intervention (from a mean of 2.9 medications at baseline). In a post-hoc analysis in medication-free patients, IOP was reduced further by 46 percent to 13.1 mm Hg at one year. There were no significant overall safety issues, nor concerns with corneal health.
“The STAR-II results at one year confirm MINIject is a remarkably effective treatment option for patients with glaucoma. It is encouraging to see that the positive six-month results have been sustained at one year,” explained Professor Julián García-Feijoó from Hospital Clinico San Carlos, Complutense University, Madrid (Spain) and a STAR-II study investigator, who presented the data at AAO.
The STAR-II trial enrolled 29 patients with open-angle glaucoma in eight centres in France, Germany and Spain. Patients will be followed for two years post-intervention.
Promising longer-term data
The one-year STAR-II data are consistent with the one-year outcomes from the first MINIject trial, STAR-I. The final results from STAR-I, which enrolled 25 patients followed-up for two years, have just been published in the peer-reviewed British Journal of Ophthalmology (BJO).1 Two year results of the STAR-I trial show sustained IOP reduction and no issues with corneal health.






